Precision antibiotics – the future treatment of infections?

by Hannah Behrens
In our Bacterial World Science Short event series, researchers present their latest findings related to themes in the exhibition. At a recent Science Short, Hannah Behrens, a University of Oxford PhD student, explained how bacteria become resistant to antibiotics and how the species-specific antibiotics she studies might reduce the worrying rise in antimicrobial resistance.
Bacteria that are resistant to antibiotics present a huge problem. I work on developing new antibiotics that will slow the development of bacterial resistance.
But let’s not get ahead of ourselves. Your body is full of bacteria. In fact, there are more bacteria than human cells in your body. Most of these bacteria are good for you; they help you digest food and protect you from diseases.
But once in a while a harmful bacterium causes an infection. This could be a lung, wound, or bladder infection, or something with a fancy name like, Black Death, tuberculosis, leprosy, syphilis or chlamydia. The doctor will then prescribe you antibiotics to kill the offending bacteria.

The development of antibiotics in the 20th century was a major breakthrough. For the first time bacterial infections could be effectively and rapidly treated. Since 1942, when antibiotics first became available, we have discovered many new antibiotics which have saved millions of lives.
However, in the last 30 years we have not managed to develop any new antibiotics. During the same time, many bacteria have adapted to become resistant to the antibiotics we do have. In 2017, a woman in the US died because she had an infection with bacteria that were resistant to all available antibiotics. It is estimated that already 700,000 people in Europe alone die because of resistant bacteria per year. What is happening?
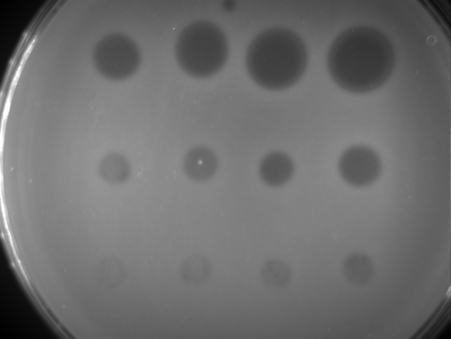
Every time we treat bacteria with antibiotics, most die, yet a few resistant bacterial cells survive. Like Rudolph the red nosed reindeer, the resistant bacteria are usually at a disadvantage until a special situation arises (a foggy night for Rudolph; treatment with antibiotics for resistant bacteria).

Under usual circumstances, producing a resistance mechanism is a disadvantage: it wastes energy and slows down growth, so very few bacteria are resistant. Only when all the non-resistant bacteria are killed by antibiotics do the resistant ones thrive. They have no more competition, and have all the resources, such as food and space, to themselves.
The more we use antibiotics, the more resistant bacteria we get. It is essential not to use antibiotics carelessly.
More antibiotics are used in animal farming than on humans. If we eat less meat, and so reduce the farming of livestock for food, we may reduce the growth of resistance bacteria. Another approach is to only take antibiotics when the doctor prescribes them. Antibiotics do not help against viral infections like colds. In many low and middle income countries, antibiotics are available in supermarkets and it is no coincidence that these countries have higher levels of resistant bacteria.

Apart from avoiding the unnecessary use of antibiotics, scientists – including me – are trying to develop better therapies against bacteria. I study precision antibiotics: drugs that specifically kill one species of bacteria. The advantage of this is that all good bacteria remain unharmed and only the disease-causing species is targeted. This also means that only resistant bacteria from this one species get an advantage to thrive.
I am interested in species-specific antibiotics against Pseudomonas aeruginosa. This bacterial species causes lung and wound infections and, according to the World Health Organization, is one of the three bacteria for which we most urgently need new antibiotics. Colleagues of mine tested different precision antibiotics against Pseudomonas and found one that is better than the others, called Pyocin S5.

I am now investigating how stable this antibiotic is, how it recognises this specific species of bacteria and how it enters the bacterial cells. This knowledge is important to decide on how to store, transport and administer the drug. I also hope that understanding why Pyocin S5 is more effective than the other antibiotics will allow us to design more effective, targeted antibiotics in the future.
My hope is that one day we will treat all bacterial infections with precision antibiotics and that antibiotic resistance will become a problem of the past.